Lyophilization Technologies is a dehydration process that takes place after a product has been frozen. Lyophilization is popular in life sciences because lyophilized reagents and assays have many benefits including ease of use and reduced total cost. Lyophilized reagents ship at ambient temperature, reduce storage and shipping costs. Lyophilized products also have a longer shelf life, leading to less waste.
Lyophilized products use less warehouse space, lower electrical and equipment costs, and reduc environmental impact. Ambient shipping is more flexible and less risky in todays complex shipping environment.
Shipments, even with critical enzymes, can be bumped off flights due to weight and dry ice. Lyophilization eliminates risk of temperature variations. No more dry ice delays. Prevent excess and obsolete inventory.
Lyophilized reagents, enzymes, beads and antibodies have been shown to be stable for years in lyophilized format. Stab-tests show antibodies >20 years old that perform like the day they were lyophilized.
Lyophilization eliminates difficult small-volume pipetting steps. Reduce user errors with simpler protocols.
Cold chain shipping adds 40x shipping material weight for domestic and 80x weight for international, compared with ambient shipping with lyophilized products! Lyophilization eliminates styrofoam coolers and dry ice. Your product ships in recyclable containers, and boosts environmental goals and regulatory compliance.
Argonaut is the exclusive producer of LyoDose beads, a proprietary spherical lyophilized bead format that contains a specified volume of reagent.






Setting up an in-house lyophilization facility requires significant investment of time and resources. Accelerate your project at a lower cost. Working with our full-service infrastructure, your project has access to:
Proprietary lyophilization technology
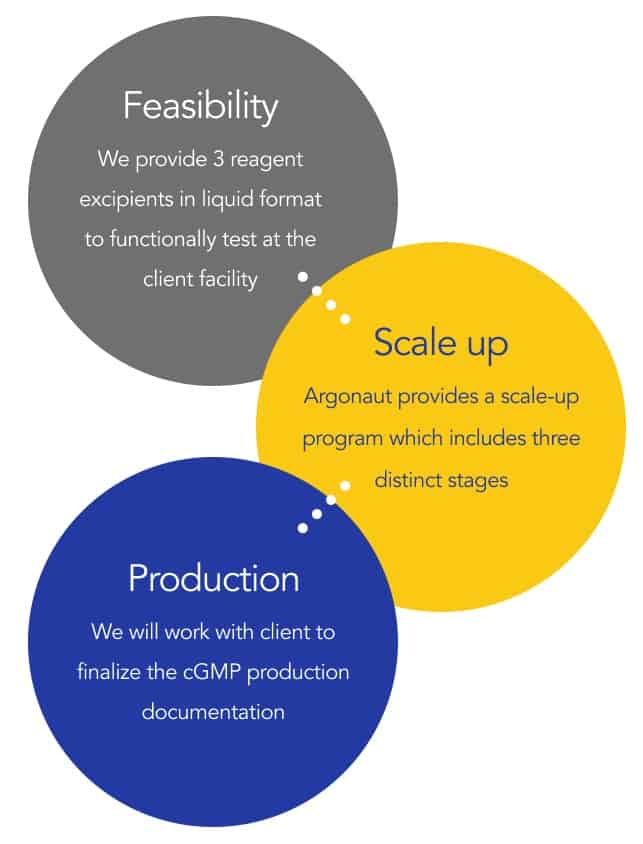
was established because development and production of lyophilized assays & reagents requires specialized expertise. Argonaut’s team can take you from liquid reagents to a fully lyophilized assay within weeks, reducing your time to market. We ensure you have the solution best matched to your goals. Click to see if Lyo is a good match!
Lyophilization is suited for small to large scale projects, for Life Sciences and Applied Markets to high-scale commercial products, and highly regulated clinical IVD products.
Download whitepaper comparing the costs of lyophilized reagents vs. wet reagents using Total Landed Cost analysis.
Many Life Science and Molecular Diagnostics companies are switching their products to a lyophilized format. But that doesn’t mean it’s the ideal match for every application.
Argonaut leads lyophilization technology but we want to ensure you have the solution best matched to your goals.
Download case studies to learn more about lyophilization and lyophilized assays, reagents & beads
Establishing a successful partnership with a CDMO requires careful planning, clear communication, and ongoing collaboration.
Here are some key steps to ensure a fruitful partnership:
Due diligence: Perform thorough research on potential CDMO partners, assessing their track record, capabilities, capacity, quality systems, and regulatory compliance. Choose a CDMO that aligns with your company’s needs and values.
Establish clear expectations: Clearly define your project goals, deliverables, and expectations at the outset of the partnership. This includes specifications for product quality, timelines, and any unique requirements.
Develop comprehensive agreements: Create detailed contracts that outline the roles and responsibilities of both parties, including intellectual property protection, payment terms, quality standards, and regulatory compliance.
Open communication: Establish and maintain open lines of communication with your CDMO partner. Regularly discuss project updates, potential issues, and any changes to the scope of work to ensure transparency and alignment.
Assign a dedicated team: Designate a project manager or team within your organization to work closely with the CDMO, overseeing project progress and ensuring that your company’s needs are met.
Engage in tech transfer: Work closely with the CDMO during the tech transfer process, sharing critical information and providing guidance to ensure a smooth transition and successful scale-up.
Monitor progress and performance: Request regular updates on manufacturing schedules, quality control results, and any challenges encountered. Use this information to make informed decisions and address potential issues proactively.
Perform audits and inspections: Conduct regular audits and inspections of the CDMO’s life science and IVD manufacturing facilities to ensure adherence to quality standards, regulatory requirements, and contract terms.
Foster a collaborative relationship: Build a strong working relationship with your CDMO partner through open communication, mutual trust, and shared goals. Work together to address any challenges that may arise and support each other’s success.
Plan for contingencies: Develop contingency plans for potential risks, such as supply chain disruptions, production delays, or regulatory changes. This can help you respond more effectively to unexpected challenges and minimize their impact.
By following these steps and working closely with a reputable CDMO like Argonaut, you can establish a successful partnership that supports your life science or IVD manufacturing needs and helps your company achieve its strategic objectives.
Maintaining control over your product development and life science or IVD manufacturing process while working with a CDMO is essential for ensuring the success of your collaboration.
Here are some key strategies to help you maintain control and stay engaged throughout the process:
Clear communication: Establish open and effective communication channels with your CDMO. Regularly discuss project updates, potential issues, and any changes to the scope of work. This ensures that both parties are aligned and helps to maintain transparency.
Establish expectations: Clearly define expectations, project goals, and deliverables at the outset of the partnership. This includes specifying quality standards, timelines, and any specific requirements for your product.
Project management: Assign a dedicated project manager or team to work closely with the CDMO, overseeing the project’s progress and ensuring that your company’s needs and expectations are met.
Detailed agreements: Develop comprehensive contracts and agreements that outline the responsibilities of each party, including intellectual property protection, quality standards, regulatory compliance, and any other relevant aspects of the collaboration.
Regular progress updates: Request regular progress reports from the CDMO, including updates on manufacturing schedules, quality control results, and any challenges encountered. This helps you stay informed and facilitates proactive decision-making.
In-person or virtual meetings: Schedule periodic in-person or virtual meetings with the CDMO to review project progress, discuss any concerns, and maintain a strong working relationship.
Conduct audits and inspections: Perform regular audits and inspections of the CDMO’s life science and IVD manufacturing facilities to verify that they adhere to your company’s quality standards and regulatory requirements. This also provides an opportunity to review their processes and make suggestions for improvement.
Active involvement: Stay actively involved in key decision-making processes, such as selecting raw materials, finalizing manufacturing processes, and approving quality control methods. This ensures that your company’s interests are well-represented throughout the collaboration.
By implementing these strategies, you can maintain control over your product development and life science or IVD manufacturing process while leveraging the expertise and capabilities of a CDMO like Argonaut. A successful partnership with a CDMO can provide significant benefits, including cost savings, access to specialized expertise, and increased capacity, while allowing your company to focus on its core competencies.
CDMOs play a crucial role in ensuring the quality of the products they manufacture and maintaining compliance with applicable regulations.
To achieve this, reputable CDMOs like Argonaut implement a comprehensive approach that includes the following elements:
Quality Management System (QMS): A robust QMS is the backbone of a CDMO’s quality and compliance efforts. The QMS includes policies, procedures, and guidelines that govern every aspect of the life science and IVD manufacturing process, from raw materials sourcing to finished product release.
Compliance with Good Manufacturing Practices (GMP): CDMOs adhere to GMP guidelines and other relevant industry standards to ensure that products are consistently manufactured according to quality requirements. GMP guidelines cover various aspects of the production process, including personnel qualifications, equipment maintenance, and record-keeping.
Regulatory expertise: CDMOs employ experienced regulatory affairs professionals who are knowledgeable about local and international regulations, industry standards, and best practices. These professionals help ensure that the CDMO stays up-to-date with the latest regulatory changes and maintains compliance throughout the life science or IVD manufacturing process.
Regular audits and inspections: CDMOs undergo regular audits and inspections by both their clients and regulatory authorities to verify that they maintain compliance with applicable regulations and quality standards. These inspections help identify potential issues and ensure that corrective actions are taken promptly.
Employee training: CDMOs invest in continuous training and education for their employees to ensure that they have the necessary skills and knowledge to maintain quality and compliance. This includes training on relevant regulations, industry standards, and best practices.
Quality control and assurance: CDMOs implement rigorous quality control (QC) and quality assurance (QA) processes throughout the life science and IVD manufacturing process. QC involves testing and monitoring of raw materials, intermediate products, and finished products to ensure that they meet established specifications. QA focuses on the overall quality management system and helps identify and address potential issues before they impact product quality.
Documentation: CDMOs maintain thorough and accurate documentation of their life science and IVD manufacturing processes, quality control results, and compliance efforts. Proper documentation is essential for demonstrating compliance with regulations and facilitating audits and inspections.
Continuous improvement: CDMOs are committed to continuous improvement in their quality and compliance efforts. They regularly review their processes, systems, and performance to identify areas for improvement and implement necessary changes.
By implementing these measures, reputable CDMOs like Argonaut can ensure that they consistently deliver high-quality products while maintaining compliance with applicable regulations. Partnering with a CDMO that prioritizes quality and regulatory compliance can provide peace of mind and help minimize potential risks associated with life science or IVD manufacturing.
The cost of outsourcing your life science or IVD manufacturing to a CDMO can vary significantly depending on several factors, such as the complexity of your product, the scale of production, the CDMO’s expertise and capabilities, and the scope of services required.
To better understand what to expect in terms of cost, consider the following factors:
Manufacturing complexity: Complex products, such as biologics, may require specialized equipment, advanced life science or IVD manufacturing techniques, or highly skilled personnel, leading to higher costs compared to simpler products.
Volume and scale: The cost per unit typically decreases as the volume of production increases, due to economies of scale. However, if your production requirements exceed the CDMO’s existing capacity, additional investments in equipment or facility expansion may be necessary, impacting the overall cost.
Scope of services: The cost of outsourcing will depend on the range of services you require from the CDMO, such as supply chain management, technical transfer, engineering development, regulatory support, or quality assurance. A more comprehensive scope of services will likely result in higher costs.
Development and tech transfer costs: The process of transferring your product’s life science or IVD manufacturing technology and know-how to the CDMO may require additional resources and time, impacting the overall cost of outsourcing.
Regulatory compliance and quality assurance: Ensuring that your product meets regulatory requirements and quality standards can be resource intensive. This includes the cost of implementing and maintaining a robust quality management system, as well as conducting regular audits and inspections.
Geographic location: The location of the CDMO’s facilities can impact the cost of outsourcing due to differences in labor costs, taxes, regulations, and logistics.
Contract terms and negotiation: The final cost will also depend on the specific terms and conditions negotiated in your contract with the CDMO. This can include factors such as payment terms, intellectual property rights, liability, and performance guarantees.
To get an accurate estimate of the cost of outsourcing your life science or IVD manufacturing to a CDMO, it is essential to clearly define your requirements and engage in a thorough evaluation and negotiation process. A reputable CDMO like Argonaut can help you navigate this process and provide a transparent, competitive, and fair pricing structure that takes into account your specific needs and expectations.
Which reagent delivery method do you prefer? We offer a wide range of reagent lyophilization services options.
Sign up to receive our newsletter & get instant access to the free Case Studies of your choice.